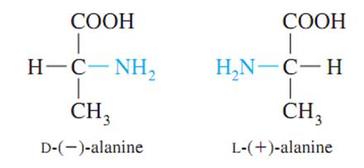Amino acid definition
Amino acids are biological molecules composed of amine (-NH2) functional group and carboxyl (COO-) functional group. These molecules are essential to our daily living. From the keratin on our finger nails to the enzymes that break down our foods.
Amino acids are amphiprotic or amphoteric molecules because they can have either positive or negative charge. Amino acids in aqueous solutions exist as zwitterions meaning they bear both positive and negative charge. The properties of amino acids depend on what substituent they have (the R group attached as side chain). Amino acids may be considered as acidic , basic, polar, or non-polar. All amino acids can either have L- or D- configuration. An amino acid has an L- configuration if it is projected in Fischer projection, the amine group is on the left side. An amino acid has a D- configuration if the placement of the amino group is on the right side if it was projected in Fischer projection. Amino acids that are classified on how the body needs them are classified into essential, nonessential, and conditional. Essential amino acids are amino acids that our body can’t produce or synthesize. They are obtained from diet. Nonessential amino acids are amino acids that our bodies produce. Conditional amino acids are those that are not required in daily living but are needed during sickness.
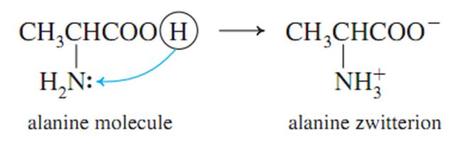
When an amino acid in solution has equal positive and negative charges, it is electrically neutral and won’t migrate to either positive or negative electrode when placed in an electrolytic cell as shown in the formula: The pH in which no migration occurs towards at either electrode is the isoelectric point.
If an acid is added to the solution containing the amino acid, its equilibrium shifts to the left (favoring the protonation of the amino group). If a base is added, the equilibrium shifts to the right (favoring the formation of carboxylate ions). |